Jiaqi Zhu, Ethan M. Rowland, Sevan Harput, Kai Riemer, Chee Hau Leow, Brett Clark, Karina Cox, Adrian Lim, Kirsten Christensen-Jeffries, Ge Zhang, Jemma Brown, Christopher Dunsby, Robert J. Eckersley, Peter D Weinberg, Meng-Xing Tang, “3D Super-Resolution US Imaging of Rabbit Lymph Node Vasculature in Vivo by Using Microbubbles“
Radiology
DOI: 10.1148/radiol.2019182593
Affiliation
J. Zhu, E. M. Rowland, S. Harput, K. Riemer, C. H. Leow, G. Zhang, P. D Weinberg and M.-X Tang are with the Department of Bioengineering, Imperial College London, United Kingdom. (e-mail: mengxing.tang@imperial.ac.uk).
B. Clark is with the Department of Imaging, Natural History Museum, London, U.K.
K. Cox is with Department of Surgery, Maidstone and Tunbridge Wells NHS Trust, Maidstone, U.K.
A. Lim is with Department of Imaging, Charing Cross Hospital, Fulham Palace Rd, London, England
K. Christensen-Jeffries, J. Brown and R. J. Eckersley are with the Division of Imaging Sciences, Biomedical Engineering Department, King’s College London, London, SE1 7EH, U.K.
C. Dunsby is with the Department of Physics and the Centre for Pathology, Imperial College London, London, SW7 2AZ, U.K.
Abstract
Background
Variations in lymph node (LN) microcirculation can be indicative of metastasis. The identification and quantification of metastatic LNs remain essential for prognosis and treatment planning, but a reliable noninvasive imaging technique is lacking. Three-dimensional super-resolution (SR) US has shown potential to noninvasively visualize microvascular networks in vivo.
Purpose
To study the feasibility of three-dimensional SR US imaging of rabbit LN microvascular structure and blood flow by using microbubbles.
Materials and Methods
In vivo studies were carried out to image popliteal LNs of two healthy male New Zealand white rabbits aged 6–8 weeks. Three-dimensional, high-frame-rate, contrast material–enhanced US was achieved by mechanically scanning with a linear imaging probe. Individual microbubbles were identified, localized, and tracked to form three-dimensional SR images and super-resolved velocity maps. Acoustic subaperture processing was used to improve image contrast and to generate enhanced power Doppler and color Doppler images. Vessel size and blood flow velocity distributions were evaluated and assessed by using Student paired t test.
Results
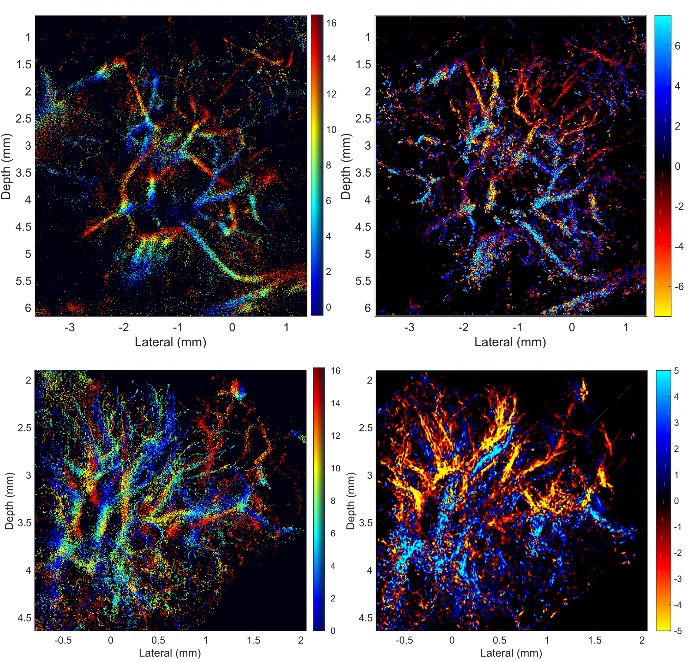
SR images revealed microvessels in the rabbit LN, with branches clearly resolved when separated by 30 µm, which is less than half of the acoustic wavelength and not resolvable by using power or color Doppler. The apparent size distribution of most vessels in the SR images was below 80 µm and agrees with micro-CT data, whereas most of those detected with Doppler techniques were larger than 80 µm in the images. The blood flow velocity distribution indicated that most of the blood flow in rabbit popliteal LN was at velocities lower than 5 mm/sec.
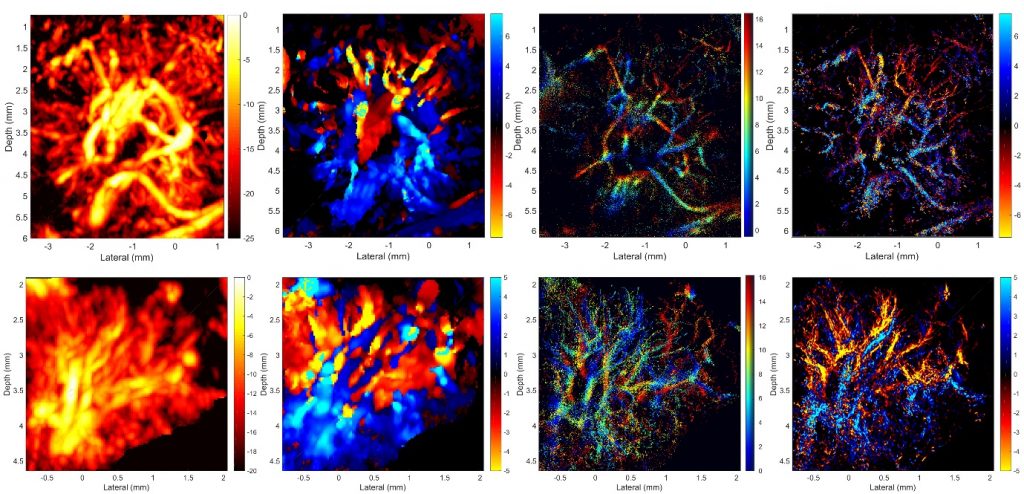
Conclusion
Three-dimensional super-resolution US imaging using microbubbles allows noninvasive nonionizing visualization and quantification of lymph node microvascular structures and blood flow dynamics with resolution below the wave diffraction limit. This technology has potential for studying the physiologic functions of the lymph system and for clinical detection of lymph node metastasis.
Supporting Bodies:
Supported in part by Engineering and Physical Sciences Research Council under grant nos. EP/N015487/1, EP/N014855/1, and EP/M011933/1; Cancer Research UK Multidisciplinary Project Award under grant no. C53470/A22353; Imperial Confidence-in-Concepts; Imperial Proof-of-Concept; and British Heart Foundation Centre of Research Excellence.
K.R. and P.D.W. supported by British Heart Foundation